As a reliable supplier of 3,5 - Difluorophenol, I am often asked about its reactivity with acylating agents. In this blog, I will delve into the reaction mechanism, reaction conditions, and applications of the reaction between 3,5 - Difluorophenol and acylating agents.

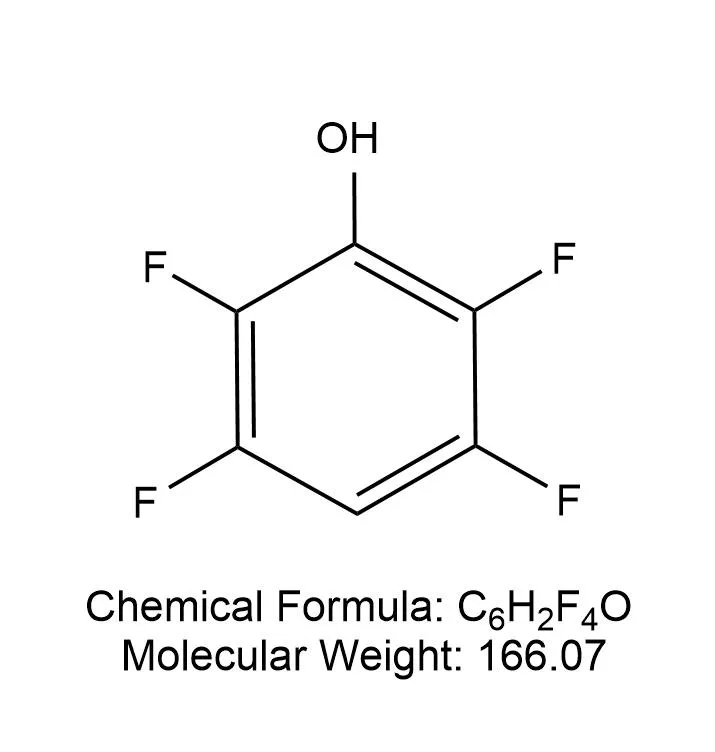
1. Introduction to 3,5 - Difluorophenol
3,5 - Difluorophenol is an important organic compound with the molecular formula C₆H₄F₂O. It has unique physical and chemical properties due to the presence of two fluorine atoms on the benzene ring. The fluorine atoms not only affect the electron density of the benzene ring but also influence the reactivity of the hydroxyl group. This compound is widely used in the synthesis of pharmaceuticals, agrochemicals, and other fine chemicals.
2. Acylating Agents
Acylating agents are compounds that can introduce an acyl group (-COR) into a molecule. Common acylating agents include acyl halides (such as acetyl chloride, benzoyl chloride), acid anhydrides (such as acetic anhydride), and esters. These agents react with nucleophiles, and in the case of 3,5 - Difluorophenol, the hydroxyl group acts as a nucleophile.
3. Reaction Mechanism
The reaction between 3,5 - Difluorophenol and acylating agents is a typical nucleophilic acyl substitution reaction. The hydroxyl group of 3,5 - Difluorophenol attacks the electrophilic carbonyl carbon of the acylating agent.
Reaction with Acyl Halides
When 3,5 - Difluorophenol reacts with an acyl halide (RCOX, where X is a halogen like Cl or Br), the reaction proceeds as follows:
- The oxygen atom of the hydroxyl group in 3,5 - Difluorophenol donates a pair of electrons to the carbonyl carbon of the acyl halide, forming a tetrahedral intermediate.
- The halogen atom (X) then leaves as a halide ion, and the tetrahedral intermediate collapses to form an ester.
The general equation for the reaction is:
C₆H₃F₂OH + RCOX → C₆H₃F₂OCOR + HX
Reaction with Acid Anhydrides
When reacting with an acid anhydride ((RCO)₂O), the mechanism is similar. The hydroxyl group attacks the carbonyl carbon of the acid anhydride, forming a tetrahedral intermediate. One of the acyl groups is transferred to the 3,5 - Difluorophenol, and a carboxylic acid is also formed as a by - product.
The general equation is:
2C₆H₃F₂OH+(RCO)₂O → 2C₆H₃F₂OCOR + RCOOH
4. Reaction Conditions
Solvents
The choice of solvent is crucial for the reaction. Polar aprotic solvents such as dichloromethane (DCM), tetrahydrofuran (THF), and acetonitrile are commonly used. These solvents can dissolve both the 3,5 - Difluorophenol and the acylating agent and also stabilize the reaction intermediates.
Catalysts
In some cases, a catalyst may be required to accelerate the reaction. Pyridine or tertiary amines (such as triethylamine) are often used as catalysts. They can react with the hydrogen halide (HX) generated during the reaction with acyl halides, shifting the equilibrium towards the formation of the ester.
Temperature
The reaction temperature depends on the reactivity of the acylating agent. For more reactive acyl halides, the reaction can be carried out at relatively low temperatures (0 - 25 °C). For less reactive acid anhydrides, higher temperatures (reflux conditions) may be required.
5. Factors Affecting the Reaction
Electronic Effects
The fluorine atoms on the benzene ring of 3,5 - Difluorophenol have a strong electron - withdrawing inductive effect. This effect decreases the electron density on the oxygen atom of the hydroxyl group, making it a slightly weaker nucleophile compared to phenol. However, the reaction still proceeds smoothly under appropriate conditions.
Steric Effects
If the acylating agent has bulky substituents, steric hindrance may affect the reaction rate. Bulky acyl groups can make it more difficult for the hydroxyl group of 3,5 - Difluorophenol to approach the carbonyl carbon of the acylating agent.
6. Applications of the Reaction Products
The esters formed by the reaction of 3,5 - Difluorophenol with acylating agents have various applications.
In Pharmaceuticals
They can be used as intermediates in the synthesis of drugs. The fluorine atoms in the molecule can enhance the lipophilicity, metabolic stability, and biological activity of the drug candidates. For example, some esters derived from 3,5 - Difluorophenol may have antibacterial or anti - inflammatory properties.
In Agrochemicals
These esters can also be used in the synthesis of pesticides and herbicides. The unique chemical structure of the products can provide specific biological activities against pests and weeds.
7. Other Fluorophenol Compounds
In addition to 3,5 - Difluorophenol, we also supply other high - quality fluorophenol compounds, such as ≥99.0% 2 - Chloro - 6 - fluorophenol CAS No.:2040 - 90 - 6, ≥99.0% 3 - Fluorophenol CAS NO.372 - 20 - 3, and 2,3,5,6 - Tetrafluorophenol. These compounds also have their own unique reactivities and applications in different fields.
8. Conclusion
The reaction between 3,5 - Difluorophenol and acylating agents is an important synthetic method for the preparation of esters. Understanding the reaction mechanism, conditions, and factors affecting the reaction is crucial for optimizing the synthesis process. The products of this reaction have wide applications in pharmaceuticals, agrochemicals, and other industries.
If you are interested in purchasing 3,5 - Difluorophenol or any of our other fluorophenol products, please feel free to contact us for procurement negotiations. We are committed to providing high - quality products and excellent customer service.
References
- March, J. Advanced Organic Chemistry: Reactions, Mechanisms, and Structure. Wiley, 2007.
- Carey, F. A., & Sundberg, R. J. Advanced Organic Chemistry Part B: Reactions and Synthesis. Springer, 2007.
- Greene, T. W., & Wuts, P. G. M. Protective Groups in Organic Synthesis. Wiley, 2007.
